If you're a frequent visitor to Healthify, why not share our site with a friend? Don't forget you can also browse Healthify without using your phone data.
Unapproved medicines
Key points about unapproved medicines
- Unapproved medicines are also called section 29 medicines.
- Approved medicines can be used in unapproved ways (also called off-label medicines).
- Read about unapproved medicines, when they are used and your right to informed consent.

- Medicines go through an assessment and approval process before they can be supplied in New Zealand. This is to ensure they meet quality standards and are safe and effective. These are known as approved medicines.
- Sometimes your healthcare professional (prescriber) may need to use an unapproved medicine or an approved medicine in a new way.
- If this is the case, the prescriber must explain to you what is being prescribed and why.
- Under the Code of Health and Disability Services Consumers’ Rights, you must also agree to it being used before it can be prescribed for you. Read more about informed consent.
- If you agree to be given an unapproved medicine, the manufacturer or importer must be advised and information sent to Medsafe. Your prescriber will tell you about this.
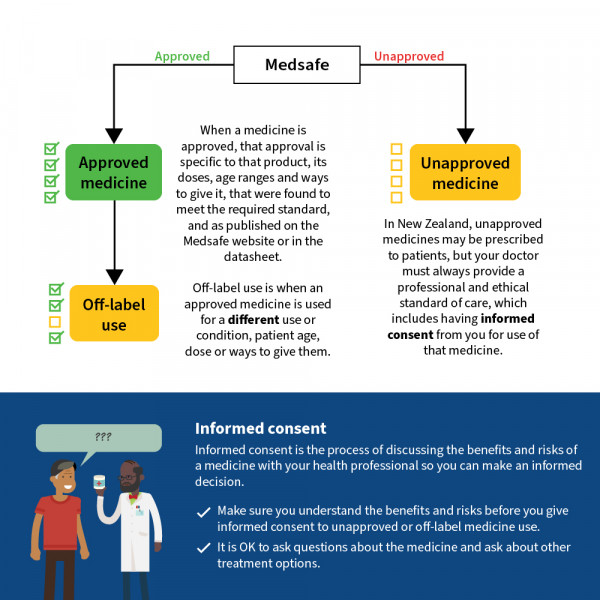
Image credit: Healthify He Puna Waiora
In Aotearoa New Zealand most medicines you are prescribed are approved medicines. This means they have been through a regulatory assessment process in New Zealand to ensure that the quality of the product meets the required standard and that the product can be considered safe and effective for the uses listed in that medicine’s data sheet.
Data sheet
A data sheet is a document that is written by the drug company for healthcare professionals and has information about the medicine, including the details for which the medicine is approved for use in New Zealand, such as the specific uses, doses, age ranges and routes (ways that a medicine is given).
Most companies also provide consumer medicine information (CMI) for patients. Data sheets and CMI for medicines are available on the Medsafe website(external link). Medsafe is the New Zealand Medicines and Medical Devices Safety Authority, and is a part of Health New Zealand | Te Whatu Ora. It is responsible for the regulation of medicines.
Before a medicine can be approved
Before a medicine can be approved, the drug company must submit clinical and scientific data to Medsafe for review. The drug company must show that the medicine is of acceptable quality and is safe and effective for its intended uses.
‘Safe’ does not mean that the medicine has no side effects. Instead, it means Medsafe has determined that the benefits of using the medicine for a particular use outweigh the potential risks overall. For some individual patients this may not be true, which is why your doctor, nurse or pharmacist considers carefully which medicines are best for you.
When a medicine is approved
When a medicine is approved, that approval is specific to the uses, doses, age ranges and routes (ways that a medicine is given) that were found to meet the required standard, and as published on the Medsafe website or in the data sheet.
If there are any changes, such as a new use or dose, this must be applied for by the drug company and approved by Medsafe.
When an approved medicine is prescribed for a different use or condition, patient age, dose or route, it is considered to be an off-label use. This means that the use, dose, age range or route has not been assessed and approved for use in New Zealand.
It is legal for doctors to use medicines off label, but they must get your informed consent first.
It’s not legal for drug companies to market (advertise or promote) their medicines for off-label uses. Off-label marketing is very different from off-label use.
If a medicine has not been approved by Medsafe it is ‘unapproved’. Unapproved medicines are also called section 29 medicines.
These are medicines (both branded and generic) available overseas that may have acceptable quality, efficacy and safety, but approval to supply these medicines has not been sought in New Zealand.
In New Zealand, unapproved medicines may be prescribed to patients, but your doctor must always provide a professional and ethical standard of care, which includes having informed consent from you for use of that medicine. Being unapproved does not mean the medicine is ‘unsafe’. Your healthcare provider will discuss the benefits and risks of the medicine with you.
There are many reasons why your doctor may consider an unapproved or off-label use of a medicine.
Prescribing medicines for children
Many medicines given to children are used off label. This is because clinical trials have not usually been done in children or the drug company has not applied for use to be approved for this age group.
Dose or route not approved in New Zealand
Sometimes medicines are prescribed off label because a non-approved dose or route is recommended or used in other countries, but the drug company has not applied for approval for this dose or route in New Zealand.
Before you are prescribed an unapproved medicine, you should be given detailed information. This should include:
- that the medicine prescribed is unapproved in New Zealand
- the possible benefits of treatment and any known risks and adverse effects
- the possibility of unknown risks and side effects
- any other available treatments using approved medicines.
If you are prescribed a medicine that is approved, but is being prescribed for a use or dose or route that is unapproved, then your doctor should also discuss this with you.
Questions you may want to consider asking your doctor
If your doctor is thinking about using an unapproved medicine or approved medicine for an unapproved use, you may want to ask them questions like these:
- What evidence supports the off-label use of this medicine to treat my medical condition?
- Is this unapproved medicine likely to work better than an approved medicine?
- Are there other medicines or therapies that are approved to treat my medical condition?
- What are the potential benefits?
- What are the risks of harm in my situation?
In New Zealand, the Medicines Act 1981 allows an authorised prescriber (such as your GP) to prescribe, administer or arrange for the administration of medicines for the treatment of a patient in their care. The medicine and its use may or may not be approved. However, only medical doctors can prescribe unapproved medicines.
Section 25 of the Medicines Act allows an authorised prescriber to “procure the sale or supply of any medicine” for a patient in their care. This means that prescribers may obtain for supply any medicine for a patient under their care (within their scope of practice), regardless of whether it is approved or unapproved in New Zealand.
However, the prescriber must always provide a professional and ethical standard of care, which includes having informed consent from the patient for use of that medicine.
Section 29 of the Medicines Act allows the sale or supply of unapproved medicines. The person or company who supplies the medicine must notify the Director-General of Health of the supply (via Medsafe), and record the prescriber, the patient and the name and place of supply. Only medical doctors can prescribe an unapproved medicine.
Approved medicines that are prescribed for an unapproved use, dose or route can be supplied or prescribed by any authorised prescriber. There is no reporting requirements associated with such supply.
Brochures
Unapproved medicine (Section 29(external link)) Pharmac 2025
Medicines and side effects(external link) Healthify He Puna Waiora, NZ, 2024
5 questions to ask about your medications (external link)Health Quality and Safety Commission, NZ, 2019 English(external link), te reo Māori(external link)
References
Brochures

Unapproved medicine (Section 29)
Pharmac, NZ, 2025

Medicines and side effects
Healthify He Puna Waiora, NZ, 2024

Health Quality and Safety Commission, NZ, 2019 English, te reo Māori
Credits: Sandra Ponen, Pharmacist, Healthify He Puna Waiora. Healthify is brought to you by Health Navigator Charitable Trust.
Reviewed by: Angela Lambie, Pharmacist, Auckland
Last reviewed:
Page last updated:





